|
Bonding electrons, which are shared by a pair of atomsĪnd nonbonding electrons, which belong to a particular atom butĭo not participate in bonding. Notice that there are two kinds of electron groups in this We canĭraw the Lewis structure on a sheet of paper. Lets consider the Lewis structure for CCl 4. Geometry is tetrahedral the bond angle is around 105 degrees. For bent molecular geometry when the electron-pair The bond angle is slightly less than 109.5 degrees, around 107ĭegrees. Geometry is trigonal planar the bond angle is slightly less thanġ20 degrees, around 118 degrees. Note: for bent molecular geometry when the electron-pair The table below summarizes the molecular and electron-pair geometriesįor different combinations of bonding groups and nonbonding pairs of electrons The Lewis structure and determine the number of bonding groups of electronsĪnd the number of non-bonding pairs of electrons on the central atom, then use 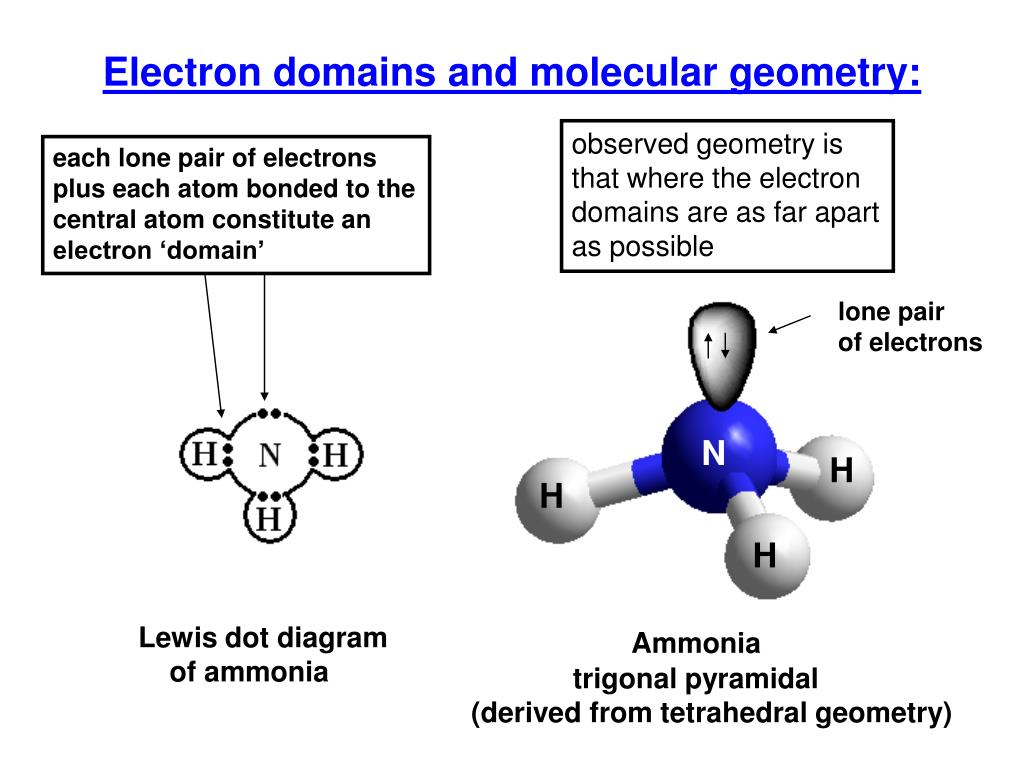
That to determine the shape (molecular geometry) of a molecule you must write Notice that there are several examples with the sameĮlectron-pair geometry, but different molecular geometries. If askedįor the electron-pair geometry on the central atom we must respond with theĮlectron-pair geometry. The shape of a molecule we must respond with a molecular geometry. The molecular geometry is the shape of the molecule. To the bond angles of between a terminal-central-terminal atom in a compound. The electron-pair geometry provides a guide Molecular geometry is the name of the geometry used Name of the geometry of the electron-pair/groups/domains on the central atom, whether theyĪre bonding or non-bonding. In this case thereĪre three groups of electrons around the central atom and the molecualr geometry When a central atom has two terminal atoms bonded by single bonds and a terminalĪtom bonded with two pairs of electrons (a double bond). The term bonding groups/domains (second from the left column) is used in the columnįor the bonding pair of electrons. 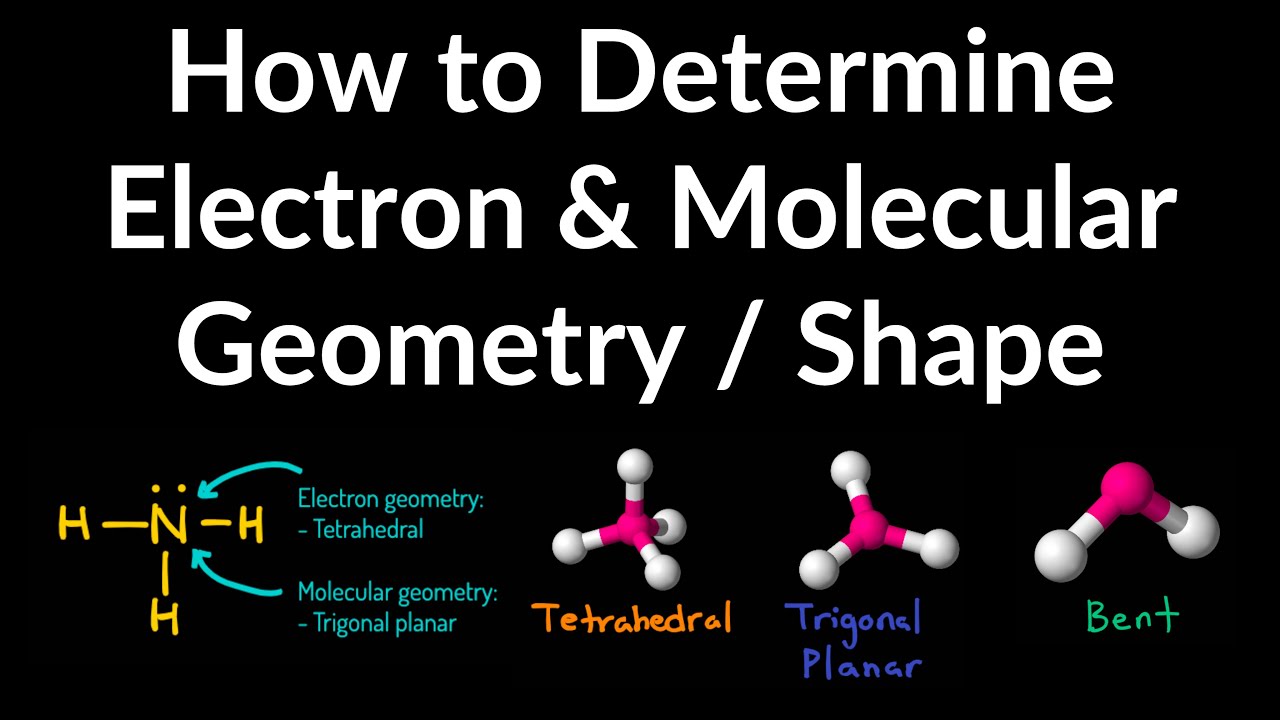
On an individual atom that are not shared with another atom. Non-bonding pairs of electrons are those pairs of electrons Of electrons are those electrons shared by the central atom and any atom to Of bonding pair of electrons and non-bonding pairs of electrons. The table below contains several columns. To use the model we will have to memorize a collection Model called the Valence Shell Electron-Pair Repulsion (VSEPR) model that isīased on the repulsive behavior of electron-pairs. 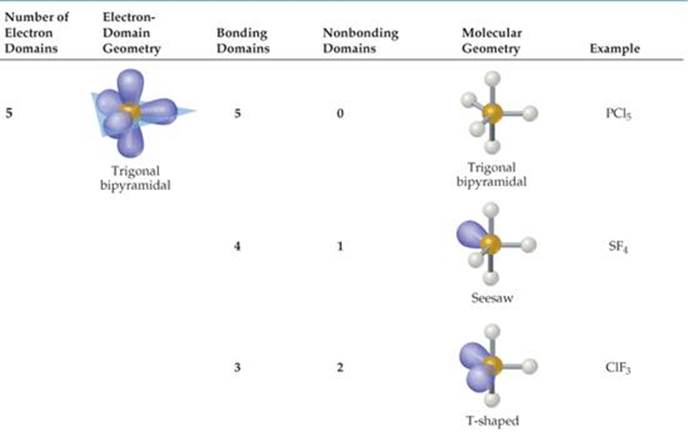
Of simple molecular (covalent) compounds and polyatomic ions. Molecular Geometry Molecular Geometry VSEPR At this point we are ready to explore the three dimensional structure
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
